Business License (Company Registration)

View LAEYO Labs’ business license and company registration details for supplier verification, including registered entity information and audit-ready documentation context.

See the factory by live video walkthrough
Book an on-site factory visit in GuangzhouUse this Trust Center to review the key evidence buyers, sourcing teams, and QA teams often request during supplier onboarding. Explore certifications and audits, compliance documents, company credentials, test reports, patents, and factory process proof. If your team needs a market-specific or SKU-specific audit pack, contact us for the relevant documents and supporting records.
Best for overseas buyer teams that want a first verification step
See production flow, cleanroom access control, QC checkpoints, and packaging areas
Review selected documentation live with our team
Best for importers, distributors, and brand teams moving toward sampling or onboarding
Visit showroom, production areas, process checkpoints, and documentation review points
Meet the team and discuss project scope face to face
Use this library to speed up supplier due diligence. Each report includes a clear title, a preview, and a short summary—request the full files for your SKU and target market.
This page helps buyers verify LAEYO as a manufacturing supplier by organizing key company credentials in one place. Review business registration, production and manufacturing licenses, institute-related qualifications, and operating records commonly requested during vendor screening, compliance review, and procurement onboarding.

View LAEYO Labs’ business license and company registration details for supplier verification, including registered entity information and audit-ready documentation context.

Zhongke Skin Gene (Guangdong) Institute of Dermatological Research is a dermatology-focused research institute engaged in skin science, ingredient efficacy evaluation, and applied research to support evidence-based skincare product development.

Official cosmetics manufacturing license for LAEYO Labs, issued by the competent authority. Covers permitted product scope and manufacturing activities, with validity details and audit-ready documentation for supplier verification.

National Enterprise Credit Information Publicity System is the official Chinese government database for verifying company registration, legal status, shareholders, and compliance disclosures. It is used for supplier due diligence and entity verification.

This license authorizes LAEYO Labs’ China entity to operate approved value-added telecommunications services in accordance with PRC regulations. It applies to online information and digital service operations

Zhongke Microview (Guangdong) Medical Research Institute conducts applied research in medical and life sciences, supporting formulation research, technical validation, and scientific assessment for industry partners.
This page helps buyers review the compliance documents commonly requested before sampling, import review, channel approval, or market entry planning. Document categories include COA, SDS/MSDS, INCI ingredient listings, IFRA-related fragrance documentation, declarations, and shipping classification records. Request the appropriate compliance pack based on your SKU, target market, and regulatory needs.
 PDF
PDF
Third-party hazard identification report confirming the listed product can be transported as non-dangerous goods, with itemized checks covering explosive risk, flammability, oxidation/organic peroxide, toxicity & infection, radiation, corrosion, and other transport hazards—supporting safer, compliant shipping documentation.
 PDF
PDF
Redacted MSDS/SDS documenting product safety and handling controls, including hazard identification, composition overview, first-aid and fire-fighting guidance, storage/handling requirements, exposure controls, disposal guidance, and transport classification.
 PDF
PDF
This Sea Freight Identification & Classification Report (DGM China, “By Sea”) assesses the listed cosmetic goods against IMDG Code criteria and confirms they are “Not subject to these regulations” for sea transport, with hazard-screening results covering flammability, oxidizing properties, toxicity/infection, corrosion, and other risks
Use this page as a visual due-diligence library for LAEYO’s cosmetic manufacturing process. Review unedited production evidence covering clean production routines, batching and emulsification, filling, packaging, and final packout. These materials help buyers verify process discipline, hygiene controls, in-process QC, and batch traceability before sampling, onboarding, or RFQ review.

A close-up of our lab emulsification step—high-shear mixing to build a stable, uniform texture before moving into evaluation and scale-up planning.

A look at our compounding area with stainless-steel mixing vessels and control panels—built for controlled processing and consistent batch production.

Automated pouch feeding and conveying on the packaging line—organized material flow designed to improve consistency, efficiency, and in-process handling control.

Inside our formulation lab: raw materials are staged and weighed on calibrated scales, then prepared in controlled steps to support repeatable prototypes and traceable development.

Unedited bench-side footage showing gift set components being placed into a custom foam insert for consistent presentation and packout—prior to overwrap/cartoning and shipment.

A quick look inside our controlled production room: operators in protective gear assemble components on a moving line with step-by-step handling and in-process checks to keep output consistent and contamination risk low.

Unedited on-site footage showing finished cartons being placed into a clear protective overwrap and sealed at the packing station—helping keep boxes clean and protected before outer-case packing and shipment.

Unedited footage from our packaging area: operators assemble and pack retail cartons on a conveyor line, supporting consistent packout and pre-shipment checks before outbound QC.

A real-time look at our emulsification step: controlled stirring and mixing to achieve uniform texture and batch consistency before filling and packaging.

A short, unedited look at our controlled packaging area: operators assemble cartons on a conveyor line under hygiene controls to support consistent packout and pre-shipment checks.

A short, unedited view of automated cartoning. This step helps keep packout consistent and reduces handling variability before outbound QC and shipment.

A quick look inside our controlled production area: stainless mixing/emulsifying tanks and operator-controlled processing.
Use this Patents and IP page to review intellectual property evidence that supports manufacturing innovation and project credibility. Each item is organized to help buyers and sourcing teams screen relevance quickly during supplier onboarding, technical review, or private label development discussions. If needed, request the full supporting files for a specific project scope.

repair composition and its preparation method and its applications

hydrating moisturizing composition and its preparation method and its applications

filling device for a ace mask filling machine

anti sensitivity repair composition and its preparation method and its applications

a vacuum drying oven for cosmetics

a rotational viscometer for laboratory use

a pure water system with sterilization and disinfection functionality

a mixing equipment for cosmetics production

a homogenizer for cosmetics laboratory use

a high pressure autoclave for cosmetic ingredient -production

a filling machine-to improve filling stability

a biochemical incubator for cosmetics
This page presents selected cosmetic test reports used to support product verification and supplier review. Reports may cover safety, microbiological limits, regulated contaminants, stability-related indicators, and other quality or performance criteria where applicable. Buyers can use this library as an initial screening step before requesting a SKU-specific report pack.
 PDF
PDF
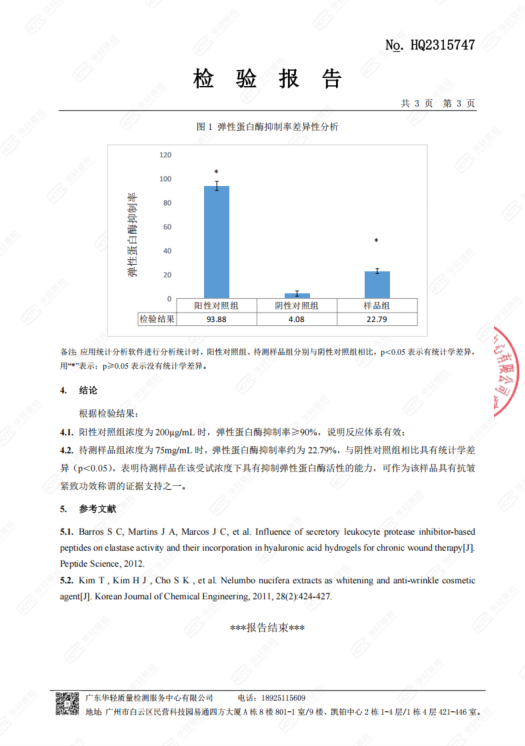
Independent lab report for “CH Proxylane Firming Cream” verifying key safety compliance items under the Cosmetic Safety Technical Specification, including microbiological limits (total count, yeast & mold, thermotolerant coliforms, S. aureus, P. aeruginosa) and chemical/contaminant limits (Hg, Pb, As, Cd, and 1,4-dioxane). Results are documented as meeting the applicable requirements for an audit-ready quality evidence […]
 PDF
PDF
Independent lab test report commissioned by LAEYO for “CH Proxylane Firming Cream”, evaluating elastase inhibition as an in-vitro indicator for anti-wrinkle/firming performance. The report provides the test method, quantified results, statistical analysis, and conclusion to support claim substantiation in an audit-ready evidence pack.
 PDF
PDF
Third-party cosmetic efficacy report documenting the test design, methodology, data charts, and conclusions for a Proxylane 30% sample, supporting repair-claim substantiation as part of our formulation validation and evidence pack
Empowering your brand with our top OEM/ODM cosmetic solutions