

See the factory by live video walkthrough
Book an on-site factory visit in Guangzhou
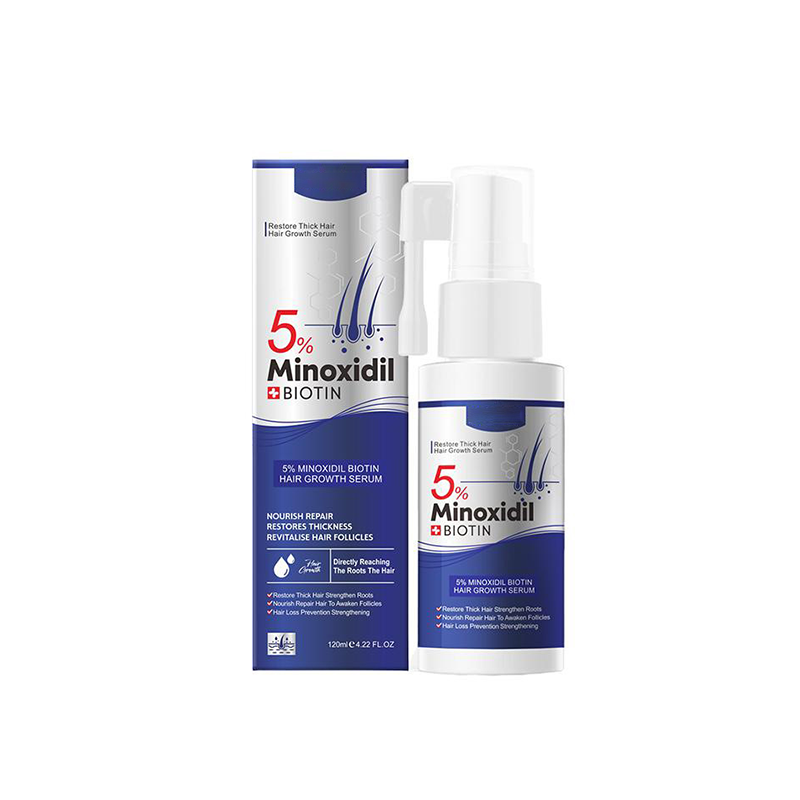
Professional-grade Minoxidil 5% and Biotin serum designed for visible hair density restoration and scalp vitality improvement.
From fast sampling to iterative optimization—aligned to your market and positioning.
Stable production workflow with quality checkpoints for consistency and scale-up.
Packaging options, decoration, labeling/INCI coordination—helping you launch shelf-ready SKUs.
thinning hair, hair loss, weak roots, daily use, all scalp types
stimulates hair growth, nourishes follicles, strengthens roots, improves scalp health, enhances density
Men and women experiencing thinning hair, postpartum shedding, or early-stage hair loss.
Ensures long-term ingredient and color stability under various temperatures.
Formulated to minimize scalp irritation; patch test recommended.
Custom efficacy test protocols can be arranged to validate hair density performance.
As your OEM partner, we accelerate sampling by starting with a pre-approved hair care formula base from our library, tailored to EU regulations like REACH and SCCS. This cuts R&D time by up to 2 weeks. We run stability, microbiological, and packaging compatibility tests in parallel during sample production, not sequentially. For EU buyers, we immediately align on critical specs: claims boundaries, ingredient restrictions, and label requirements to avoid rework. Packaging is sourced from pre-tooled components or rapid prototyping to fit samples quickly. We provide documentation support including CPSRs and test reports, but final regulatory approval is your responsibility as the brand owner. The golden sample process typically takes 3-4 weeks with expedited options, versus 6-8 weeks standard. Key drivers: clear briefs, using existing molds, and upfront compliance checks. We recommend a pilot batch of 500-1000 units post-sample to validate scale-up before full production.
European buyers compare hair care suppliers by first assessing EU compliance support. They look for suppliers who can assist with safety assessments, product information files (PIF), and testing for heavy metals, allergens, and microbial limits under Regulation 1223/2009. This includes documentation management and guidance on labeling requirements. For QA and consistency, buyers review the supplier's quality systems, such as in-house microbiology labs, stability testing, and batch uniformity checks for viscosity, pH, and performance. Consistency is ensured through standardized production and regular audits. Concrete details: MOQ guidance typically starts at 5,000 units for shampoos and conditioners. Sampling timeline is 2-4 weeks for formula approval. Lead time from production to shipping is 8-12 weeks, depending on packaging complexity. Packaging must comply with EU labeling, including INCI lists and multilingual options. We support all necessary testing and documentation but cannot guarantee regulatory approval; buyers should verify with local authorities.
To get an accurate hair care quote for the EU market, include your target channels like Amazon, retail, or salons, and list all SKUs with formats such as shampoo, conditioner, or mask and sizes like 200ml or 500ml. Specify packaging materials and designs, noting if you need sustainable options like PCR plastic or refillable systems, as EU compliance requires specific labeling and material safety. Also, outline any claims like sulfate-free or color-safe, as we can help ensure they meet EU Regulation (EC) No 1223/2009 through documentation and testing. Provide your budget per unit and launch timeline to align production. MOQs typically start at 3,000-5,000 units per SKU but drop with higher volumes or simpler packaging. Sampling takes 4-6 weeks with stability and compatibility checks, while full production lead time is 8-12 weeks post-approval, including CPNP notification and safety assessment support.
To formulate a scalp serum for oily scalp without irritation, we focus on lightweight, non-comedogenic bases such as hyaluronic acid or squalane, and incorporate actives like zinc PCA for sebum regulation and prebiotics for microbiome balance. We exclude potential irritants like alcohol denat or fragrances, and conduct patch testing during R&D to minimize risk. For sampling, we offer lab samples within 2-3 weeks, with MOQs typically starting at 10,000-20,000 units per SKU depending on packaging complexity. Common packaging includes airless pumps or dropper bottles to prevent contamination and ensure precise application. Testing involves microbial analysis, stability studies under EU storage conditions, and compatibility checks with packaging materials. We support documentation like the Product Information File (PIF) and safety assessments, but do not guarantee regulatory approval. Lead time from final sample approval to production is 8-12 weeks, factoring in raw material sourcing and quality control stages. Our GMP-certified facility ensures consistent batch quality for EU market entry.
In the EU, anti-dandruff shampoos must be positioned as cosmetics, not drugs, by focusing on cosmetic claims like 'helps reduce visible flakes' and using approved ingredients such as piroctone olamine or climbazole, while avoiding banned substances like zinc pyrithione in certain concentrations. Key details include MOQ guidance of 10,000 units minimum, which can drop to 5,000 for stock packaging, and a sampling timeline of 3-4 weeks after formula approval, including stability checks for viscosity and pH under varied temperatures. For testing and docs, we conduct microbial hold-time tests, provide a Product Information File (PIF) with safety assessments, and ensure packaging labels meet EU regulation (EC) No 1223/2009 with ingredient lists in local languages. Lead time from production to shipping is 8-12 weeks, factoring in batch QA for leakage risks and pigment dispersion in final mixes. We avoid guaranteeing regulatory approval but assist with all necessary compliance steps to streamline your launch.
In the EU, shelf life and Period After Opening (PAO) for hair care products are established based on EU Cosmetics Regulation (EC) No 1223/2009, which requires evidence of product stability over time. We conduct accelerated stability testing at elevated temperatures (e.g., 40°C for 3 months) and real-time testing under normal conditions to determine expiration dates and recommend PAO symbols (like 6M or 12M) based on formulation type and packaging. For MOQ guidance, minimum order quantities of 5,000-10,000 units often justify the cost of full stability studies, with smaller batches relying on accelerated data but requiring longer lead times. Sampling timelines typically involve 4-6 weeks for initial stability checks, followed by 3-6 months for complete testing cycles, depending on product complexity (e.g., shampoos vs. serums). Testing and documentation include stability reports, microbial challenge tests to assess preservative efficacy, and compatibility tests with packaging materials to prevent leaching or degradation. We assist in preparing all necessary documents, such as Product Information Files (PIFs) and safety assessments, for EU market entry, though final regulatory approval is the brand's responsibility. Packaging choices, like airless pumps or UV-protected bottles, can extend shelf life by reducing oxidation, and we optimize production lead times to 8-12 weeks from final approval to shipping, incorporating QA checkpoints for batch consistency.
A Certificate of Analysis (COA) is a mandatory quality document for hair care products like shampoo, conditioner, hair mask, and scalp serum in the EU. It confirms each batch meets specified standards under EU Regulation EC No 1223/2009, ensuring traceability and safety for consumers. Without a proper COA, products cannot be legally sold or imported into the EU market, risking non-compliance penalties or recalls. Key inclusions are product name and code, batch number, production and expiry dates, manufacturer details, and test parameters with acceptance limits. For hair care, typical tests cover pH (4.0-7.0 for most), viscosity, microbial limits (e.g., <100 CFU/g for bacteria), heavy metals (lead, arsenic), preservative efficacy, and stability data under accelerated conditions. The COA must also state compliance with EU cosmetic product safety reports and ingredient restrictions, such as banned substances or allergen labeling. From a factory perspective, generating a COA involves batch-specific testing after production, which adds 1-2 weeks to lead time depending on lab capacity. MOQ influences this: smaller batches may have higher per-unit testing costs, while larger batches ensure consistency and cost efficiency. Sampling for COA requires pilot batches to validate formulations, with timelines of 3-4 weeks for testing and documentation. Packaging must be validated for compatibility to prevent leaching or contamination, affecting COA stability claims. We support by conducting in-house or third-party tests, providing draft COAs for review, and assisting with documentation for EU notifications, but final regulatory approval depends on authorities. Always factor in COA preparation when planning launch dates to avoid delays.
For EU shipments of private label hair care, the compliance document pack must meet Regulation (EC) No 1223/2009. Key elements are the Product Information File (PIF) with a safety assessment by an expert, detailed formula with ingredient concentrations, and proof for claims like 'volumizing' or 'repair'. We start this during sampling, which takes 2-3 weeks after formula approval, and MOQ guidance is typically 5,000 units per SKU to justify documentation costs. Testing includes microbiological, stability, and packaging compatibility checks to ensure safety and prevent leakage. Packaging must have EU-compliant labels with INCI ingredient lists in the target country's language, and we provide GMP certificates. Lead time from order to shipment is 8-10 weeks, including 2 weeks for document compilation. We support with all tests and documentation, but brands must verify final compliance with local authorities. For efficiency, we recommend batching orders to optimize MOQ and reduce lead time variations, and our QA team aligns in-process checks with documentation.